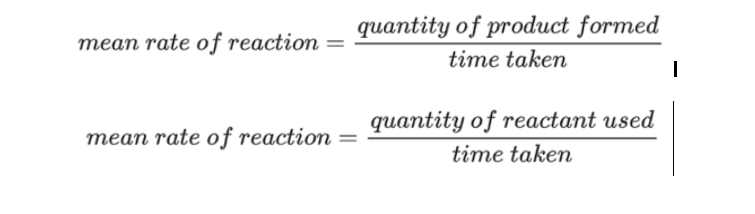Enrich your knowledge with our informative blogs
How to Calculate The Rate of Reaction in Biology?

The Rate of reaction is a measure of knowing how quickly a reactant is utilized or a product is formed. The primary focus is to determine the Rate of reaction.
However, there are several ways to determine the rate of reaction. The chosen method usually depends on the products and reactants involved and how easily these changes can be measured.
The reaction rate can also be said as the speed at which a reaction proceeds towards equilibrium. A reaction rate is generally governed by the energy barrier between products and reactions. In general, we can say that energy must be added to the reactants in order to overcome the energy barrier, and this added energy is referred to as activation energy.
The Rate of reaction can be calculated by using either of these equations:
Visualize the in-depth understanding of the natural world!
Biology would sound more interesting when your curiosity levels are satisfied with better visuals & logical explanations.
Categories
Recent Posts
- List of the qualities you should look for in your tutors?
- What is the most useful formulas in math?
- Describe the process of eating to defecation of food?
- Difference between the natural and artificial active response by the immunology system.
- Explain the different circle theorems
- How are nerve cells adapted to their function?