Enrich your knowledge with our informative blogs
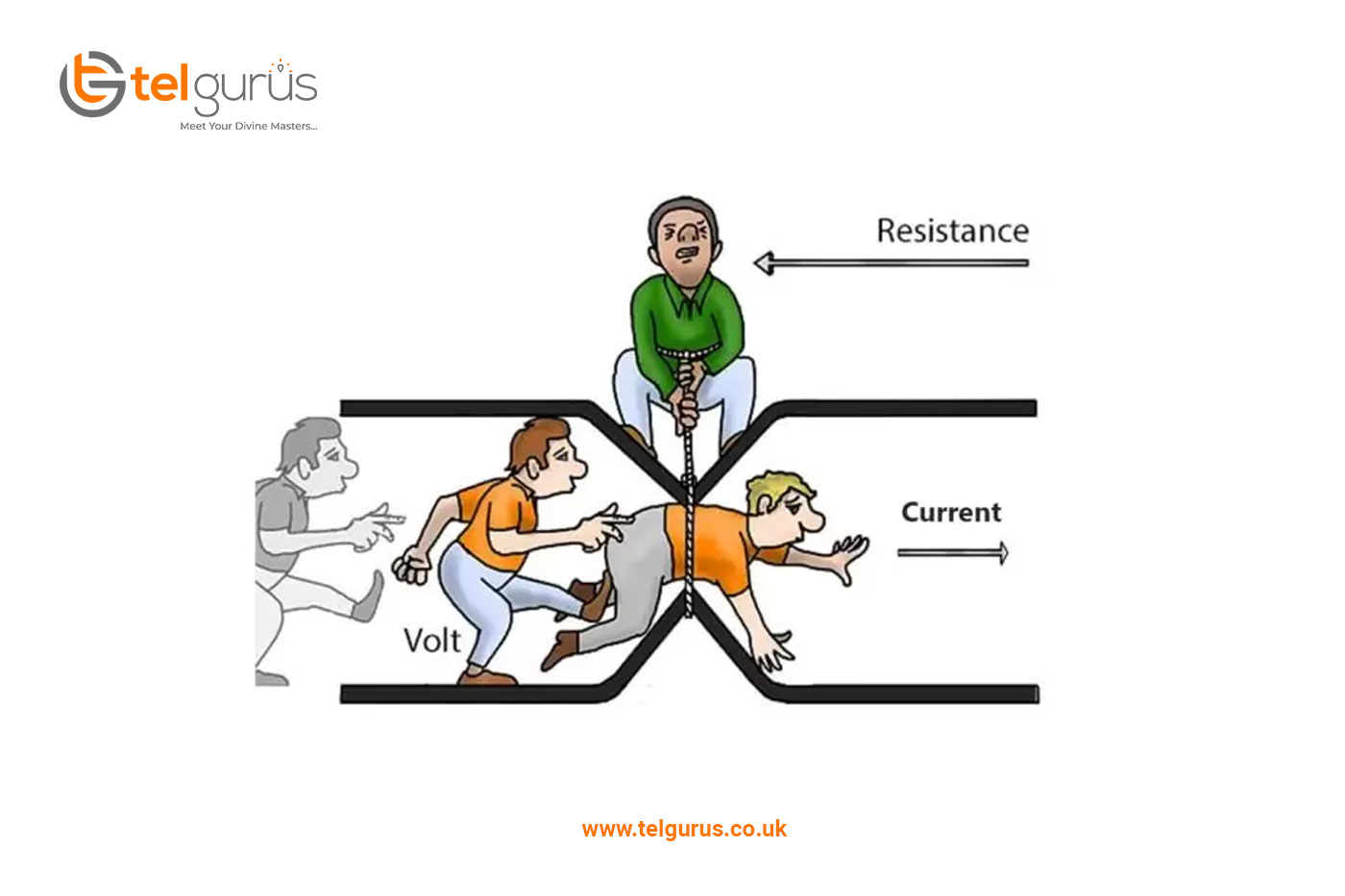
What is the Law of energy conservation?

Energy is defined as the capacity to do work and is also required to evolve life forms on earth. You must have heard about the several forms of Energy, including heat, chemical, electrical, etc. Several principles and laws govern Energy which is known as the Law of Conservation of Energy. Let us dive into the laws of conservation of Energy.
What does the Law of conservation of energy state?
The Law of Conservation of Energy states that “Energy can neither be created nor destroyed. It may only be transformed from one form to another.”
This states that a system always has a constant amount of Energy unless the Energy is added from outside.
While taking all forms of Energy into account, an isolated system’s total Energy always remains constant. Every form of Energy follows the Law of conservation of energy and states that “In a closed system, the total energy of a system is conserved.”
In an isolated system like the universe, if there is some energy loss in some part, then there also must be the gain of an equal amount of Energy in some other part.
The energy amount in any isolated system is determined by:
UT = UI + W + Q
Where
- UT stands for the total Energy of a system
- UI stands for initial Energy of a system
- Q represents the head removed or added from the system
- W is the work done on or by the system
The change in internal Energy of a system is determined by
ΔU=W+Q
This equation is also a statement of thermodynamics first law.
About Energy Conservation
Energy conservation is not only about confining the resources utilization that will finally run out. The ideal conservation method would be diminishing demand on a determined supply and enabling that supply to initiate rebuilding itself. Ample times one best way of performing this is by replacing the Energy utilized with an alternative.
Examples of Law of Conservation of Energy
Ample mechanical and electrical devices function on the Law of Conservation of Energy. Here are a few examples of the Law of Conservation of Energy.
- In hydroelectric power plants, water falls over the turbines from a certain height and rotates the turbine, producing electricity. Therefore, the water’s potential Energy is converted into the turbine’s kinetic Energy that is further converted into Electrical Energy.
- In a torch, the battery’s chemical energy is converted into an electrical one that is further converted to light and heat energy.
- In a microphone, the sound Energy is transformed into electrical Energy.
- In a loudspeaker, the electrical Energy is transformed to sound Energy.
- When the fuels are burnt, the chemical Energy is then transformed into light and heat energy.
- In a generator, mechanical Energy is transformed into electrical Energy.
- The chemical energy from a food item is transformed to thermal Energy when it is broken down in the body and is utilized to keep it warm.
Read More – Physics Questions
View More – Useful links for Your Child’s Development

Accelerate your learning in the right Dimension!
Instant doubt resolution and practical exposure to concepts, makes learning Physics easy and fun.
Categories
Recent Posts
- List of the qualities you should look for in your tutors?
- What is the most useful formulas in math?
- Describe the process of eating to defecation of food?
- Difference between the natural and artificial active response by the immunology system.
- Explain the different circle theorems
- How are nerve cells adapted to their function?