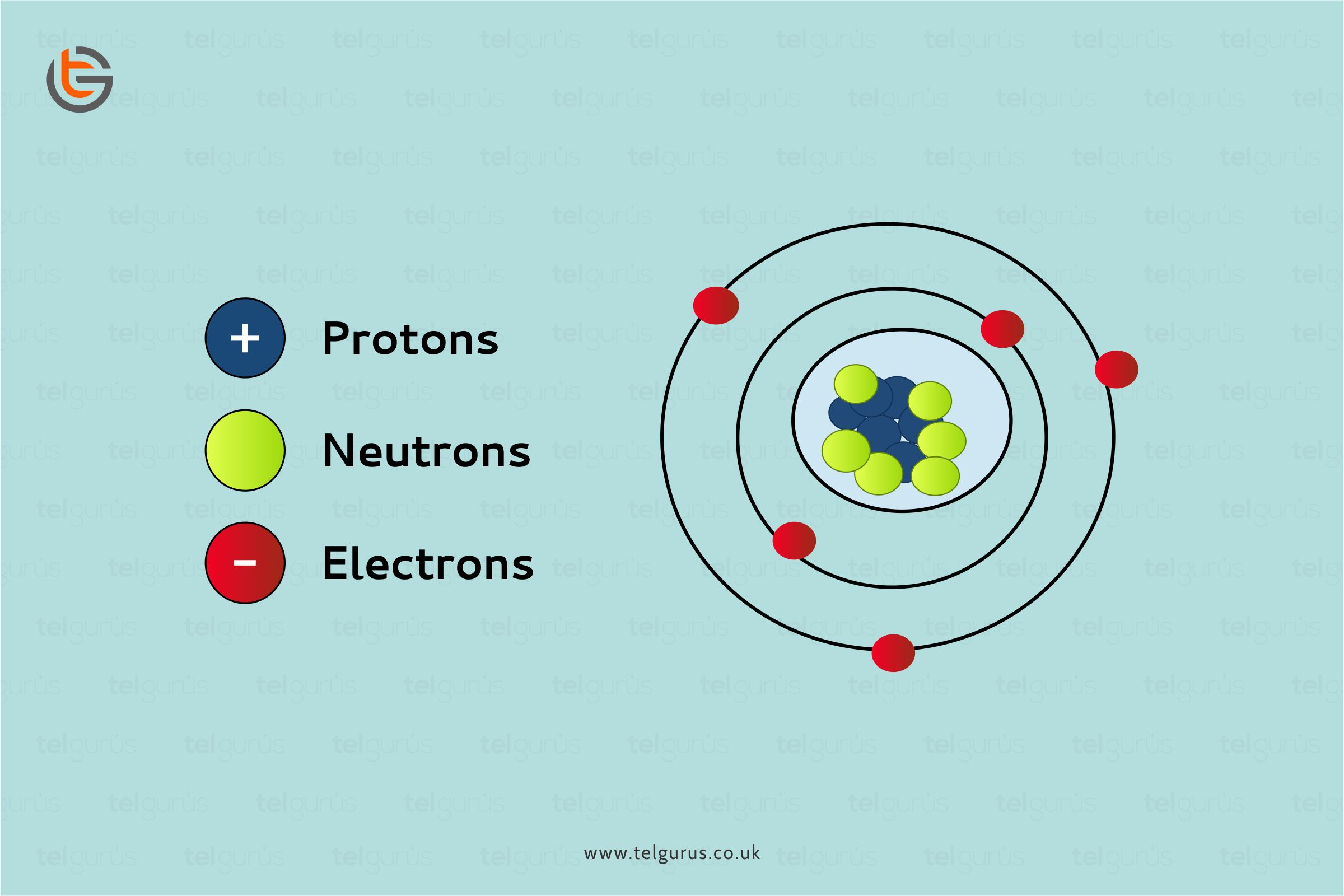
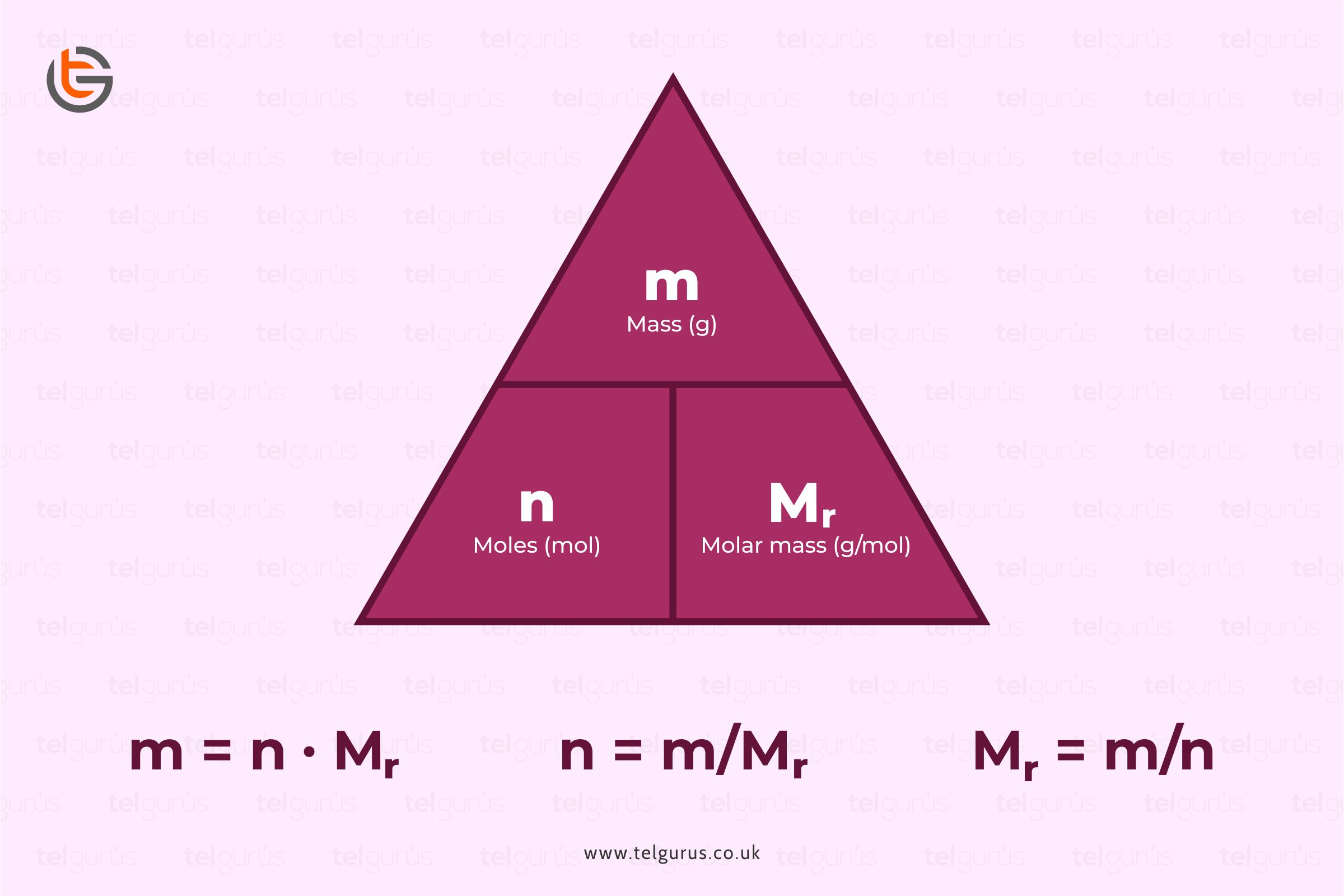
Enrich your knowledge with our informative blogs
How To Balance Equation Chemistry?
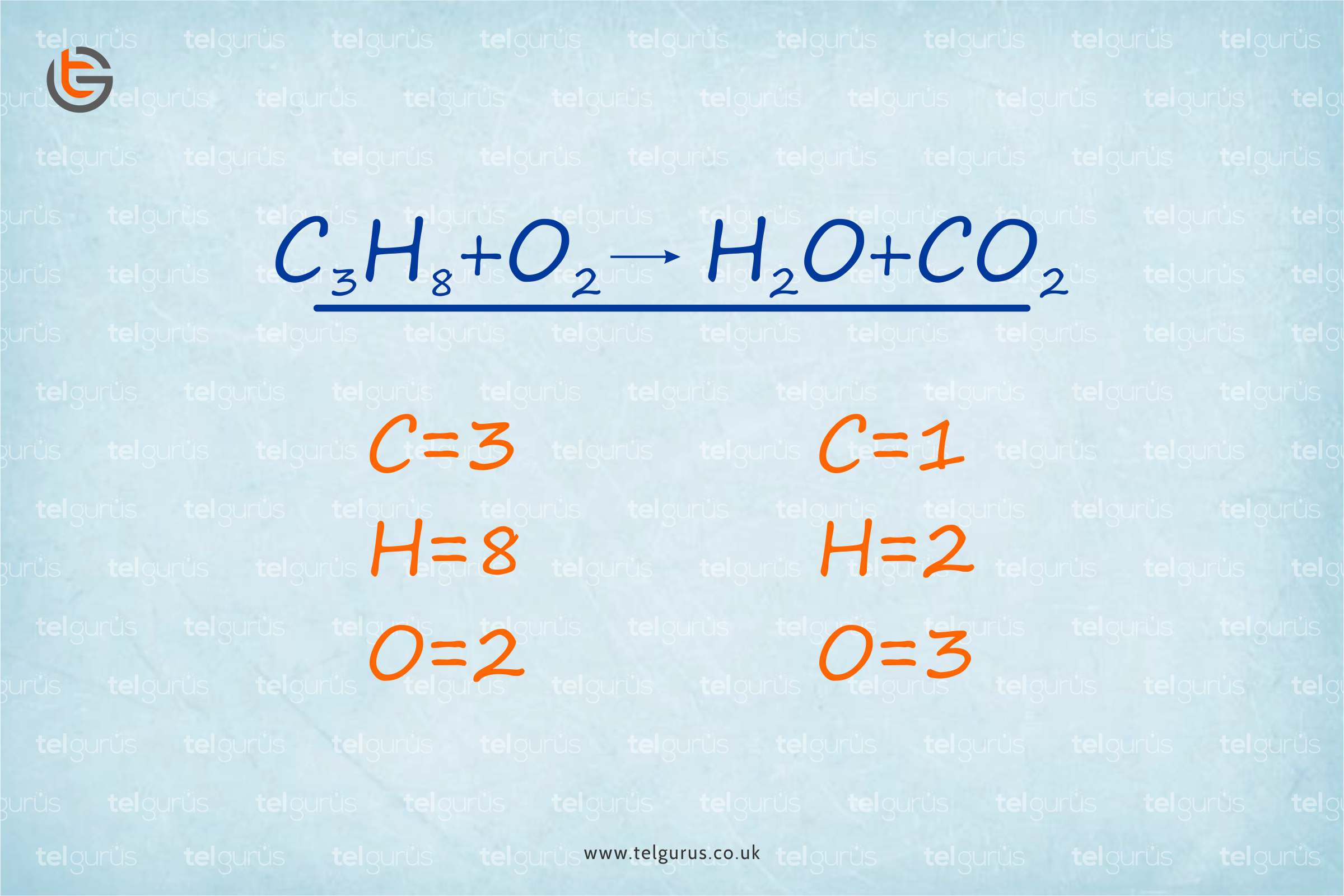
Balancing a chemical equation includes the addition of stoichiometric coefficients to products and reactants. The balancing of chemical equations is essential as a chemical equation must obey the law of constant proportions and the law of mass conversation. Balancing an equation refers to the same number of atoms of each element that must exist on both the reactant and the product side of an equation.
If you have an unbalanced equation, the chemical equation must be balanced, and for this, the balancing methods must be followed.
A chemical equation refers to a chemical reactions’ symbolic representation where their respective chemical formulas denote the products and reactants.
For instance,
Let us take a chemical equation,
2H2 + O2 ————-> 2H2O
Here in this reaction, the left-hand side is the reactant side, and the right-hand side is the product one.
Stoichiometric Coefficient
A stoichiometric coefficient describes a chemical species’ total number of molecules which take part in the chemical reaction. Like in the above equation, the stoichiometric coefficient of H2 and H2O is 2, whereas O2 is 1.
Balancing the equation!
To balance an equation, follow the step-by-step procedure, and you will never feel any complications in balancing the equation.
STEP 1
The chemical equation is
C3H8 + O2 ———–> CO2 + H2O
Now that the equation is unbalanced, we need to balance it.
STEP 2
The total number of each element’s atoms on the product and reactant side need to be compared.
Reactant side Product Side
3Carbon atoms from C3H8 1 Carbon atom from CO2
8 Hydrogen atoms from C3H8 2 Hydrogen atoms from H2O
2 Oxygen atoms from O2 3 Oxygen atoms, 1 from H2O and 2 from CO2
Now that the number of atoms is compared, perform step 3
STEP 3
Here, the stoichiometric coefficients are added to the molecules comprising an element with a different number of atoms in both the reactant and product side. The stoichiometric coefficients must balance the number of atoms on each side.
C3H8 + O2 ———–> 3CO2 + H2O
Reactant Side Product Side
3 Carbon atoms from C3H8 3 Carbon atoms from CO2
8 Hydrogen atoms from C3H8 2 Hydrogen atoms from H2O
2 Oxygen atoms from O2 7 Oxygen atoms, 1 from H2O and 6 from CO2
The Carbon atoms are equal now.
STEP 4
The above step must be repeated until the reactant, and product side’s total number of atoms becomes equal for each element.
C3H8 + O2 —–> 3CO2 + 4H2O
Reactant Side Product Side
3 Carbon atoms from C3H8 3 Carbon atoms from CO2
8 Hydrogen atoms from C3H8 8 Hydrogen atoms from H2O
2 Oxygen atoms from O2 10 Oxygen atoms, 4 from H2O, and 6 from CO2
Now the Hydrogen atoms are also balanced.
STEP 5
Balancing the Oxygen atoms
C3H8 + 5O2 ——–> 3CO2 + 4H2O
Reactant Side Product Side
3 Carbon atoms from C3H8 3 Carbon atoms from CO2
8 Hydrogen atoms from C3H8 8 Hydrogen atoms from H2O
10 Oxygen atoms from O2 10 Oxygen atoms, 4 from H2O, and 6 from CO2
Oxygen atoms are also balanced.
STEP 6
Now that all the individual elements are balanced, you just need to write down the final equation and compare it all once again.
C3H8 + 5O2 ———-> 3CO2 + 4H2O
All the elements are equal. So, this is your final balanced equation.
Read More – Chemistry Questions
View More – Useful links for Your Child’s Development

Discover the exact logic behind the reactions!
Get a deeper understanding of every possible interaction between atoms, molecules and elements in an easy and fun-loving way.
Categories
Recent Posts
- List of the qualities you should look for in your tutors?
- What is the most useful formulas in math?
- Describe the process of eating to defecation of food?
- Difference between the natural and artificial active response by the immunology system.
- Explain the different circle theorems
- How are nerve cells adapted to their function?