Enrich your knowledge with our informative blogs
How to calculate moles in chemistry?
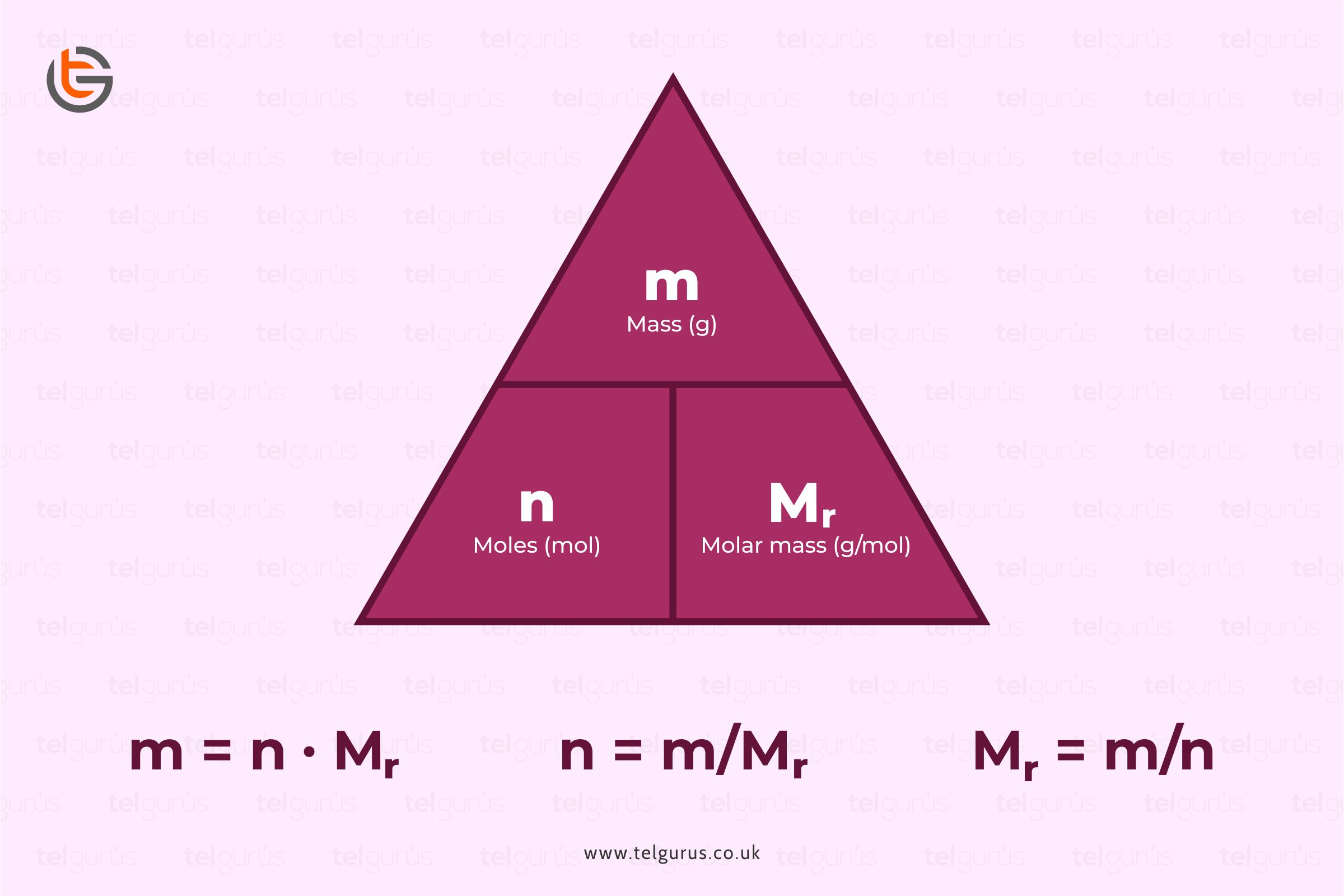
A mole is defined as a substance’s mass that comprises an equal quantity of basic units. The unit of the mole is denoted by mol.
One mole of any substance is typically equal to the value of Avogadro Number, i.e., 6.023 X 1023
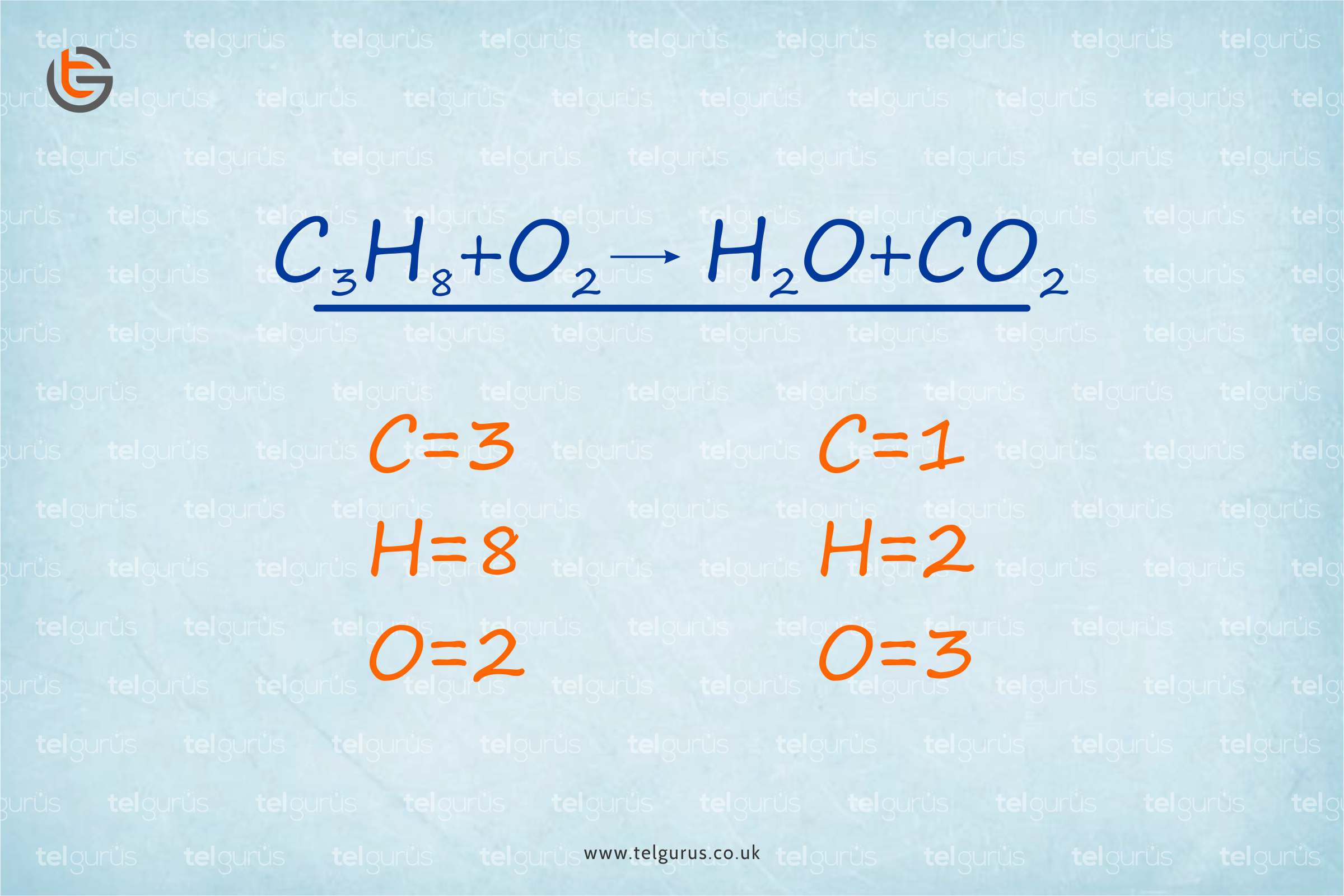
Coming to calculating the moles in chemistry, let us take an example to get a clear idea.
Number of moles = Mass of a substance / Mass of one mole
For instance
Determine the number of moles in 95 grams of MnO2
Given that, mass of MnO2 = 95 g
And the Mass of one mole MnO2 = 86.94 g
The number of moles can be calculated as
The number of moles = Mass of a substance/ Mass of one mole
Putting the values,
Therefore, Number of moles = 95/ 86.94 = 1.092 mol
Read More – Chemistry Questions
View More – Useful links for Your Child’s Development

Discover the exact logic behind the reactions!
Get a deeper understanding of every possible interaction between atoms, molecules and elements in an easy and fun-loving way.
Categories
Recent Posts
- List of the qualities you should look for in your tutors?
- What is the most useful formulas in math?
- Describe the process of eating to defecation of food?
- Difference between the natural and artificial active response by the immunology system.
- Explain the different circle theorems
- How are nerve cells adapted to their function?