Enrich your knowledge with our informative blogs
WHAT IS CO IN CHEMISTRY?
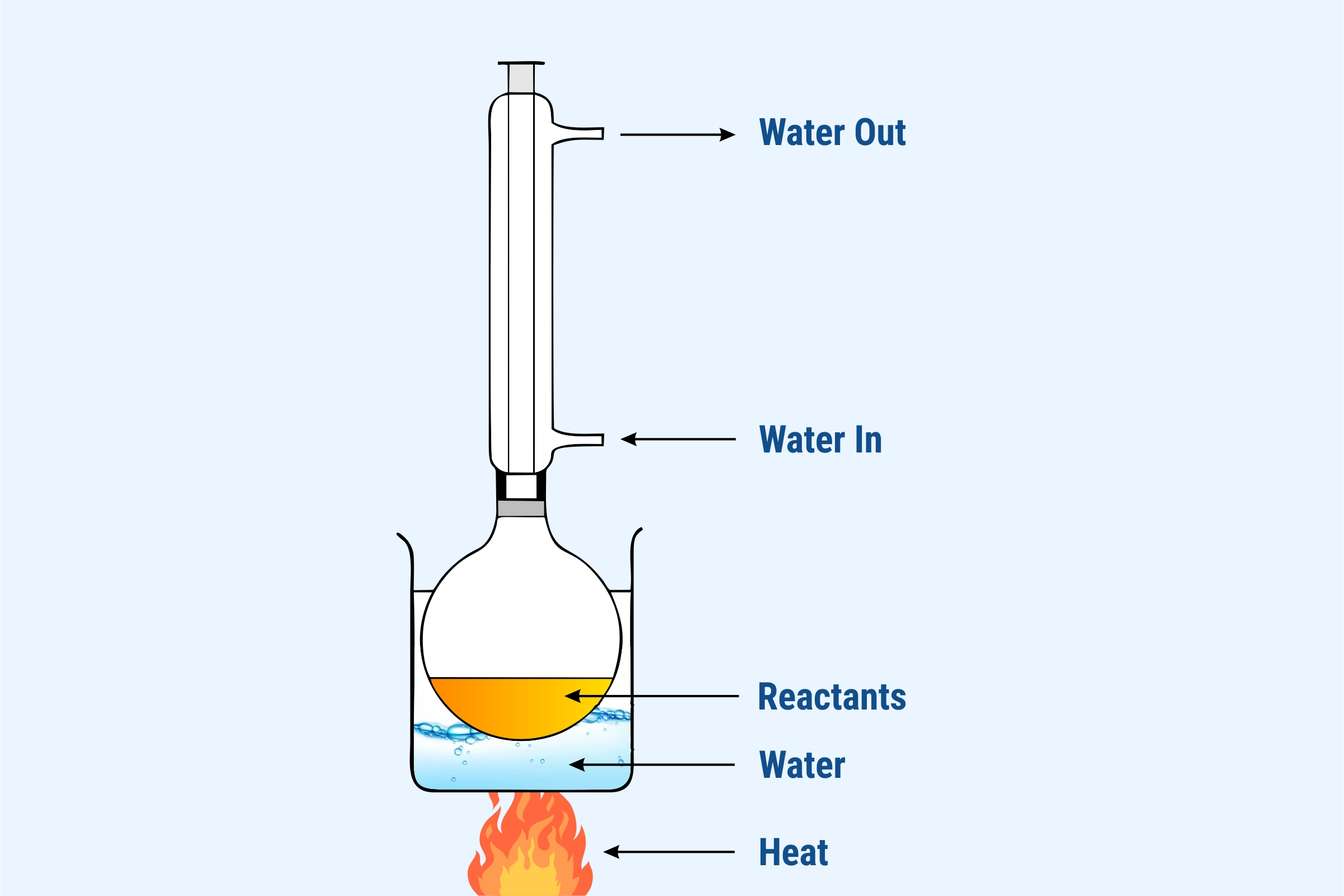
CO in chemistry is a combination of two atoms i.e. One atom of Carbon and one atom of Oxygen. Both are attached by two covalent bonds and has a single dative bond. Thus, on reacting the two atoms a new element CO, known as ‘Carbon Monoxide,’ is formed.
- The compound is formed by reacting Carbon and Oxygen at feverish temperature by adding a catalyst.
- It has a molar mass of 28.01 g per mole.
- It is highly toxic and has no colour and odour.
Uses of CO
- The main usage of CO is seen in the production of methanol.
- Carbon monoxide is a strong reducing agent.
- It is used in food materials to make them more acidic.
- CO can clear out the rust settled on the surfaces of metals.
Book Your 60-minutes Free Trial class NOW!

Discover the exact logic behind the reactions!
Get a deeper understanding of every possible interaction between atoms, molecules and elements in an easy and fun-loving way.
Categories
Recent Posts
- List of the qualities you should look for in your tutors?
- What is the most useful formulas in math?
- Describe the process of eating to defecation of food?
- Difference between the natural and artificial active response by the immunology system.
- Explain the different circle theorems
- How are nerve cells adapted to their function?